CIBMTR 50th Anniversary

Throughout 2022, we celebrated the 50th anniversary of CIBMTR® (the Center for International Blood and Marrow Transplant®). Cellular therapy has come a long way from its roots in the Cold War of the 1950s, from an experimental treatment for radiation sickness to a standard therapy for leukemia, lymphoma, and sickle cell disease. Now, cellular therapy includes both blood and marrow transplantation and other adoptive cellular therapies.
The story began in 1972 when a handful of doctors from the US, France, and the Netherlands created an international registry of transplants to see which therapies helped people the most. Over time, this informal alliance involved many more countries and a formal research collaboration between the Medical College of Wisconsin (MCW) and NMDP.
By 2022, CIBMTR had received data from about 350 medical centers worldwide and analyzed clinical outcomes of 600,000 people who had undergone blood and marrow transplantation. CIBMTR had also expanded to collect data for other cell therapies, such as chimeric antigen receptor T cell (CAR-T) therapy.
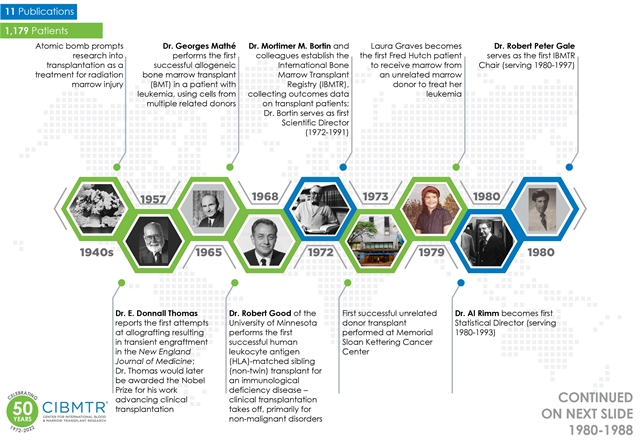
To mark the 50th anniversary, CIBMTR developed a timeline, capturing some key moments from the past 50 years. Click on the image below to view the timeline.
An important note about the timeline:
The choice of papers for this timeline is somewhat arbitrary and meant to illustrate how CIBMTR has evolved as a research organization and highlight areas where CIBMTR studies have particularly impacted the understanding and practice of hematopoietic stem cell transplantation and cellular therapy. It is not an exhaustive history of the field of BMT. Also, while some key individuals are acknowledged, including our Advisory Committees Chairs, CIBMTR's success results from the contributions of thousands of people committed to collaborating to answer important questions. These include the many experts who have participated in CIBMTR's Advisory and Working Committees and Task Forces; the dedicated physicians and research staff at participating sites who provide the data that fuel these studies; the medical, statistical, information technology, administrative, and clinical research staff at the Medical College of Wisconsin and NMDP who collaborate with the scientific community to manage and analyze CIBMTR data; the federal, non-profit, and corporate sponsors who support this work; and, importantly, the hundreds of thousands of patients who have agreed to share their data to benefit those coming after them.
Thank you for celebrating 50 years of CIBMTR—we look forward to the next 50 years!